II. The Chemistry of Life (Chapter 2: Background for Biochemistry)
A. Key Forces Acting on Matter
B. Matter
|
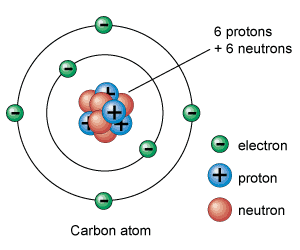
C. The AtomThe Quantum Mechanical Model of the atom is the current theory of the atom that states that it is impossible to locate the exact location of an electron. The model is created from quantum theory. The quantum theory says matter has properties associated with waves.
D. Sub-Atomic Particles
|
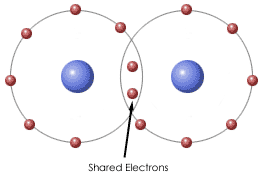
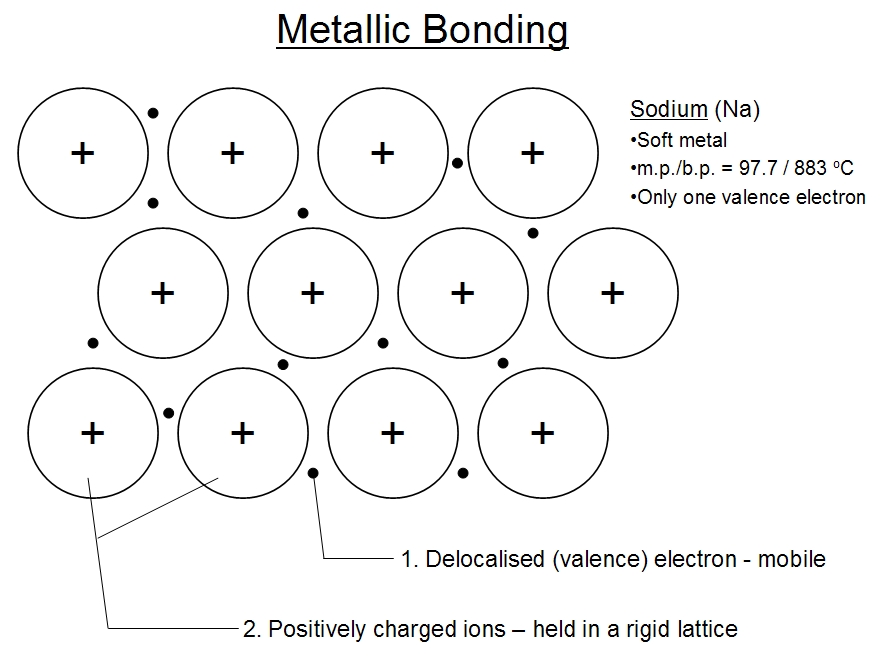
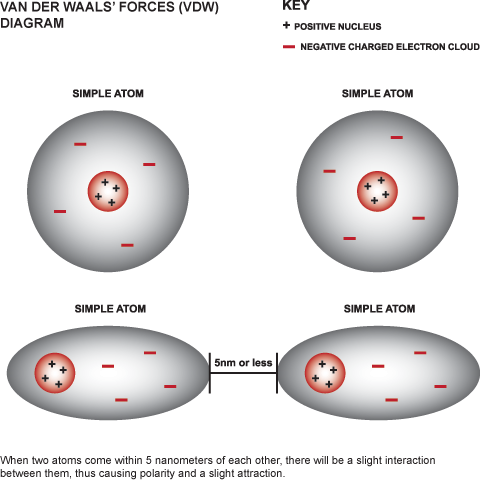
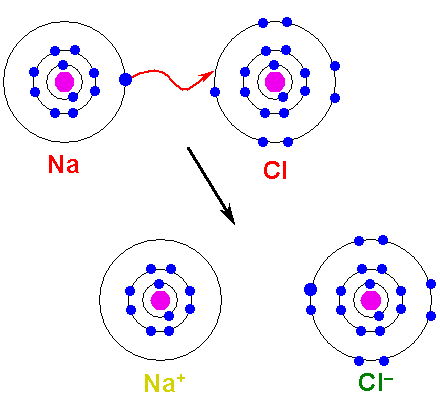
E. Chemical BondsTypes
|
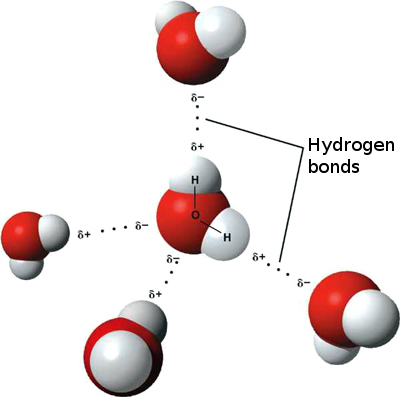
E. Chemical Bonds ImagesF. Water and It's Importance to Life 1. Water's Molecular Structure
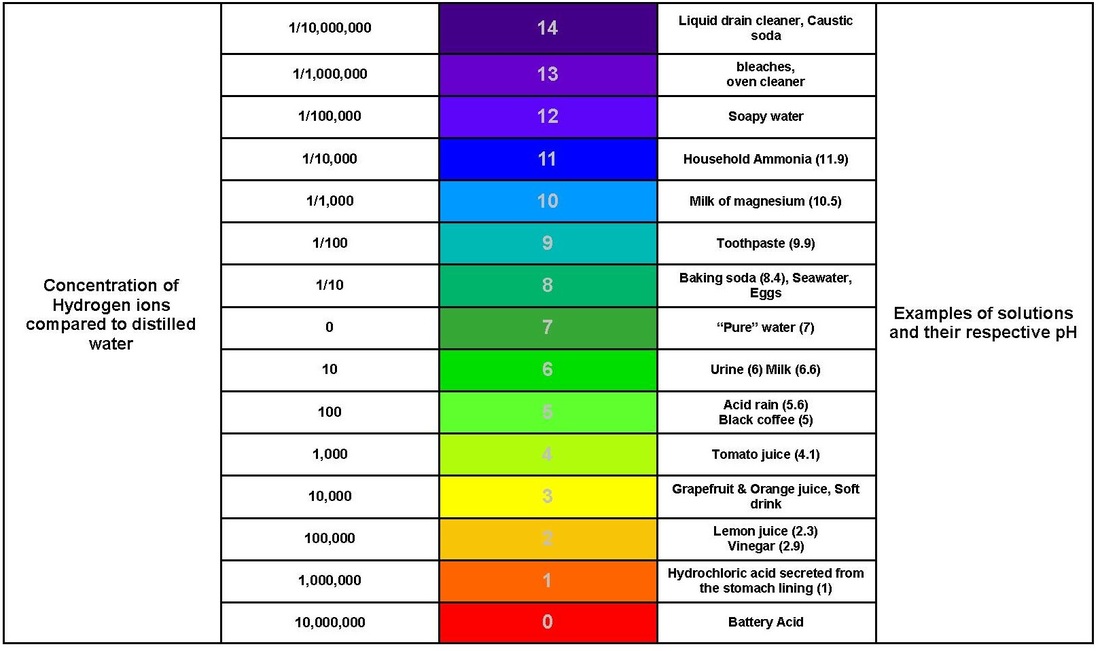
Water is a polar molecule because oxygen is very electronegative (the amount of attraction for bonding electrons) and oxygen attracts hydrogen atoms. Polarity is the property of having distinct and opposite poles. Adhesion is the force of attraction between two different molecules or the binding of two cells. The process of two different objects sticking to the surface of each other. 2. Water in Solutions and Suspensions Water is the universal solvent because more substances get dissolved in water than in any other chemical. Solute: A minor component in a solution that is dissolves by the solvent. Solvent: A substance that is used to dissolve the Solute. Suspension are particles in a substance mixed together. The particles in suspensions are larger that the ones in solutions. 3. Acids, Bases and pH The pH scale is a measurement where a substance is measured by the strength of pH (the amount of hydrogen ions in the solution). Acids are a compound that forms H+ ions in solutions. Bases are a compound that forms OH- ions (hydroxide ions) in solutions. |