Enzyme-Controlled Reactions
A. ProblemThe purpose of this lab is to observe how enzymes act on substrates to produce products. We will observe how enzyme activity is altered when the enzyme environment's pH is changed.
B. Hypothesis
|
C. Materials
D. Procedure
|
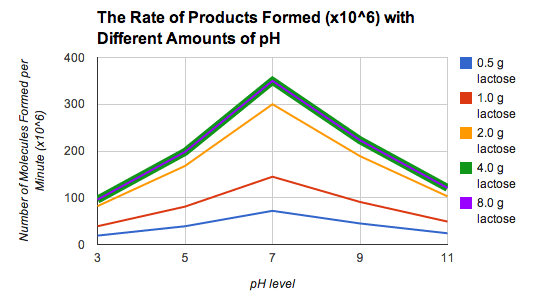
E. ResultsFor trial 1, the products formed increased as the pH continued until a level of pH 7. The products began to decrease after pH 7. For trials 2 through 4, the results were similar to trial 1. In trial 5, the results were the same with trial 4. There was no change.
|
The Rate of Products Formed (x10^6) with Different Amounts of pH |
F. Conclusion
Virtual Lab> Journal Questions
The objective of this lab is to find how pH will effect enzymes forming products. If the pH of the lactase is more acidic, the enzymes will form a lot less products. If the pH of the lactase is more basic, the enzymes will create less products than in neutral pH. A graph trend shows an increase of products formed near pH level 7 and the decrease of products formed near pH levels 3 and 11. I learned that if lactase and enzymes form products in an optimal pH, the highest amount of products will be formed as shown as the trend of the 5 trials on the graph. Lactase likes pH 7 as evidenced by the product amount formed in pH 7 showing the highest rate of product formation always being greatest at pH level 7 for every trail using different concentrations of lactose. If enzymes are not at an optimal pH, the amount of products formed will not be as high compared to enzymes in a pH level of 7 since their active site may denature which does not allow the formation of products because the enzyme-substrate complex cannot be created. Strong acidic pH creates less products than strong basic pH. From trial 1, the number of products formed in pH 3 is 19 (10^6) while the number of products formed in pH 11 is 24 (10^6). If the number of lactose grams increases past 4.0, the amount of products formed will not change as the products formed in 4.0 and 8.0 grams of lactose are 96, 198, 350, 221, 121 from pH 3 to pH 11. The trend in the graph is that the amount of products increase until a pH level of 7, then the amount of products decrease after a pH of 7. The trend occurs in all 5 of the trials. The initial rate of product formation does not increase when adding more substrates because there will be not enough enzymes to react with the extra substrates. I would improve the amount of enzymes and the amount of grams lactose given to find if there would be a change in the trend on the graph. I feel that the lactose are too low because some amounts of lactose are too difficult to duplicate. An unavoidable error in this lab would be adding the correct amount of lactose with pH because adding exactly 0.5 grams is very difficult to create. The commonalities of the results are that the different levels lactose have a similar relationship with the increase and decrease of products.
- The relationship between substrates and the rate of the reaction is that is that the initial rate will only increase if there are enough enzymes for the substrates.
- The maximum initial reaction rate will be 350 products formed per minute at pH 7.
- The maximum initial reaction rate cannot be reached at low concentrations because there are not enough substrates to react with enzymes.
- The optimal level of pH for this reaction should be at pH 7.
- The importance of temperature suggests that regulating temperature can affect enzymes that will also effect the products.
- The producer of the product should be aware that stomach is acidic. The maker should be careful that the stomach will not destroy the enzymes. The enzymes in the pill should still have the ability to make products after consumed.
The objective of this lab is to find how pH will effect enzymes forming products. If the pH of the lactase is more acidic, the enzymes will form a lot less products. If the pH of the lactase is more basic, the enzymes will create less products than in neutral pH. A graph trend shows an increase of products formed near pH level 7 and the decrease of products formed near pH levels 3 and 11. I learned that if lactase and enzymes form products in an optimal pH, the highest amount of products will be formed as shown as the trend of the 5 trials on the graph. Lactase likes pH 7 as evidenced by the product amount formed in pH 7 showing the highest rate of product formation always being greatest at pH level 7 for every trail using different concentrations of lactose. If enzymes are not at an optimal pH, the amount of products formed will not be as high compared to enzymes in a pH level of 7 since their active site may denature which does not allow the formation of products because the enzyme-substrate complex cannot be created. Strong acidic pH creates less products than strong basic pH. From trial 1, the number of products formed in pH 3 is 19 (10^6) while the number of products formed in pH 11 is 24 (10^6). If the number of lactose grams increases past 4.0, the amount of products formed will not change as the products formed in 4.0 and 8.0 grams of lactose are 96, 198, 350, 221, 121 from pH 3 to pH 11. The trend in the graph is that the amount of products increase until a pH level of 7, then the amount of products decrease after a pH of 7. The trend occurs in all 5 of the trials. The initial rate of product formation does not increase when adding more substrates because there will be not enough enzymes to react with the extra substrates. I would improve the amount of enzymes and the amount of grams lactose given to find if there would be a change in the trend on the graph. I feel that the lactose are too low because some amounts of lactose are too difficult to duplicate. An unavoidable error in this lab would be adding the correct amount of lactose with pH because adding exactly 0.5 grams is very difficult to create. The commonalities of the results are that the different levels lactose have a similar relationship with the increase and decrease of products.